Department of Redox Molecular Medicine,
Tohoku University Graduate School of Medicine
 東北大学大学院医学系研究科レドックス分子医学分野
東北大学大学院医学系研究科レドックス分子医学分野
Research Projects
Research Projects
Chemistry of Supersulfides
Sulfur, a Group 16 chalcogen element, can adopt multiple oxidation states and forms chains of sulfur atoms through a unique property known as catenation. As a result, various sulfur metabolites such as persulfides (RSSH) and polysulfides (RSSnR) are generated in biological systems (Fig. 1).
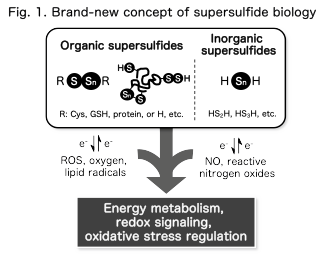
These molecules possess stronger nucleophilicity and redox activity than classical sulfur species such as hydrogen sulfide and thiols. Through modification of protein cysteine residues and participation in electron transfer reactions, they regulate diverse cellular processes including signaling pathways, oxidative stress responses, energy metabolism, and immune regulation. We collectively define these reactive sulfur species as supersulfides and investigate their chemical properties and biological functions.
Supersulfide Omics: Metabolic Analysis of Supersulfides
Because supersulfides are highly reactive and unstable, their quantitative analysis in biological systems had long been challenging. Our laboratory has developed original mass spectrometry–based analytical methods and established supersulfide metabolomics, enabling comprehensive analysis of endogenous supersulfide species (Akaike et al., Nat Commun, 2017; Fig. 2).

This platform allows us to characterize supersulfide metabolic dynamics in cells and tissues and to reveal changes in sulfur metabolic networks under physiological and pathological conditions. We are applying this approach to various disease models to better understand the physiological roles of supersulfide metabolism.
Endogenous Production of Supersulfides
Recent studies have demonstrated that supersulfides are enzymatically produced in biological systems. We have shown that several enzymes, including aminoacyl-tRNA synthetases, can generate persulfides and polysulfides from cysteine (Akaike et al., Nat Commun, 2017; Fig. 3).
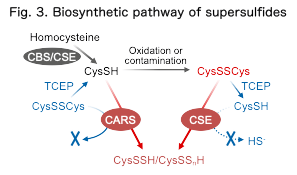
Our current research focuses on elucidating the molecular mechanisms of these enzymatic reactions and understanding how supersulfides are produced and function within cellular metabolic networks.
Sulfur Respiration in Mammals
Sulfur respiration, in which sulfur compounds function as electron acceptors, is well known in microorganisms. Emerging evidence suggests that supersulfides may also participate in electron transfer reactions in mammalian cells and influence mitochondrial function and energy metabolism (Fig. 4).
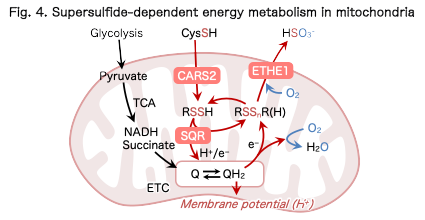
We investigate how supersulfides interact with the mitochondrial electron transport chain and regulate cellular energy metabolism and redox balance, with the goal of establishing the concept of sulfur respiration in mammals.
Supersulfide Catalysis and Ferroptosis Regulation
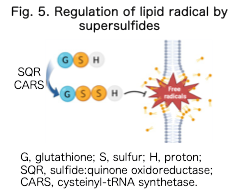
Supersulfides possess strong nucleophilic and reducing properties and are thought to regulate lipid peroxidation and electron transfer reactions (Kasamatsu et al., Sci Adv, 2023). Ferroptosis, an iron-dependent form of regulated cell death, has recently been implicated in various diseases. We are exploring how supersulfides regulate ferroptosis through their roles in redox-catalytic reactions and lipid peroxidation (Barayeu et al., Nat Chem Biol, 2023; Fig. 5).
Supersulfides in Hypoxia, Infection, and Inflammation
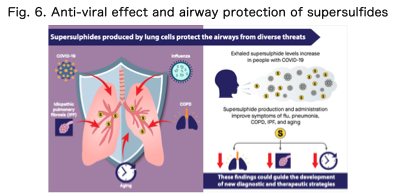
Pathological conditions such as infection, inflammation, and ischemia often involve both hypoxia and oxidative stress. However, the redox molecular networks that regulate these responses remain incompletely understood. Our laboratory investigates how supersulfides modulate hypoxic stress and inflammatory responses, aiming to uncover new redox regulatory mechanisms involved in infectious and inflammatory diseases (Fig. 6).
Breath Omics for Non-Invasive Diagnostics
We are developing breath omics, a technology for comprehensive analysis of metabolites present in exhaled breath (Fig. 7). Breath is a readily accessible and non-invasive biospecimen that reflects systemic metabolic states.
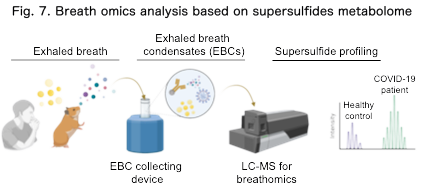
Our studies have shown that supersulfide metabolites and other redox-related molecules in breath change under disease conditions. In particular, we reported for the first time that characteristic alterations in exhaled supersulfide metabolites reflect the infection status of COVID-19 patients (Matsunaga et al., Nat Commun, 2023). We are extending this approach to cancer, chronic inflammatory diseases, and aging-related conditions to develop next-generation non-invasive diagnostics.
Advanced Analytical Infrastructure and Industry Collaboration
Our research is supported by advanced analytical platforms centered on mass spectrometry, enabling highly sensitive detection of supersulfides and trace molecules in biological samples and breath. Through close collaboration with analytical instrument manufacturers, we have improved analytical methodologies and visualized molecular species that were previously difficult to detect. In April 2024, the Shimadzu–Tohoku University Co-Creation Research Center for Supersulfide Life Science was established to promote collaborative research on supersulfides in aging, disease diagnostics, therapeutics, and functional food development (Fig. 8).


information店舗情報
東北大学大学院医学系研究科
レドックス分子医学分野
〒980-8575
宮城県仙台市青葉区星陵町2-1
TEL.022-717-8164
FAX.022-717-8219
→アクセス

